Bentrio® demonstrating strong efficacy in 2-week seasonal allergic rhinitis study
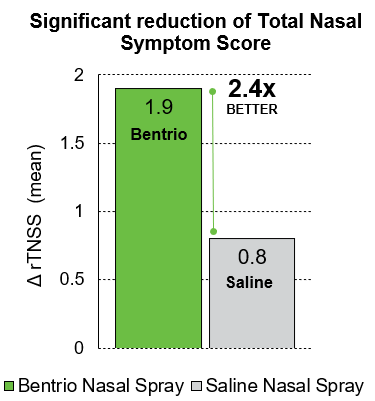
In a randomized controlled clinical trial with 100 patients suffering from seasonal allergic rhinitis in Australia, Bentrio® showed a strong reduction in the reflective Total Nasal Symptom Score (rTNSS) over a two-week treatment period. The rTNSS refers to the worst level of nasal congestion, sneezing, nasal itching, and rhinorrhea over the past 24 hours. Patients who treated themselves with Bentrio®, reported on average a decrease in the rTNSS from 6.9 points at baseline to 5.0 points (i.e. -1.9 points). Study participants who treated themselves with saline nasal spray, the current standard of care in drug-free allergy treatment, showed a decrease from 6.9 to 6.1 points (i.e. -0.8 points). The reduction in nasal symptoms conferred by Bentrio® was thus 2.4 times larger than with saline nasal spray, statistically significant (p = 0.013; 95% confidence interval -2.0 to -0.2) and clinically relevant.
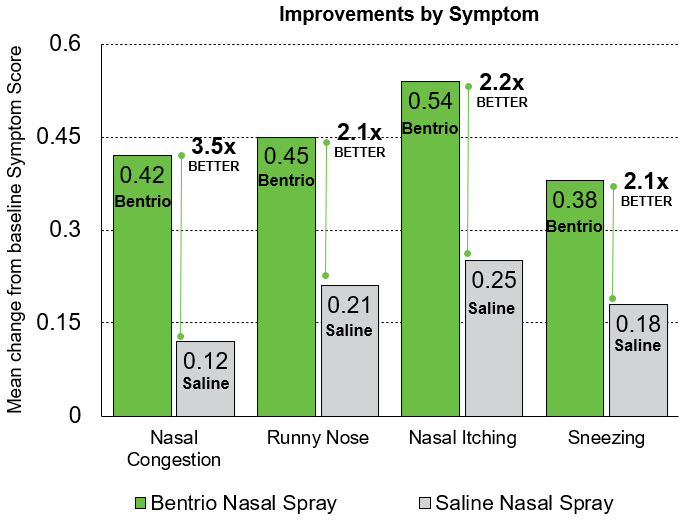
In the NASAR clinical trial, Bentrio® Nasal Spray significantly outperformed saline nasal spray, the current standard of care in drug-free allergy treatment, improving symptoms 2.1 to 3.5-times better. Patients reported their symptoms — nasal congestion, sneezing, nasal itching, and rhinorrhea — using the reflective Total Nasal Symptom Score (rTNSS)
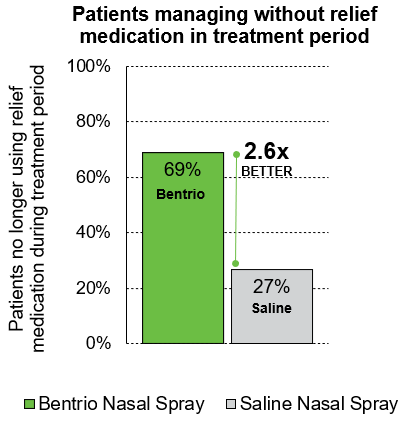
Participants in the NASAR trial were allowed to take cetirizine nasal spray as relief medication as needed. Whereas a similar share of patients required relief medication during the treatment-free run-in period, 2.6 x more patients in the Bentrio group (11 out of 16; 69%) could abstain from relief medication during treatment period (4 out of 15; 27% in saline spray group). This data points to Bentrio’s effectiveness in reducing the need for medicated allergy relief medication.
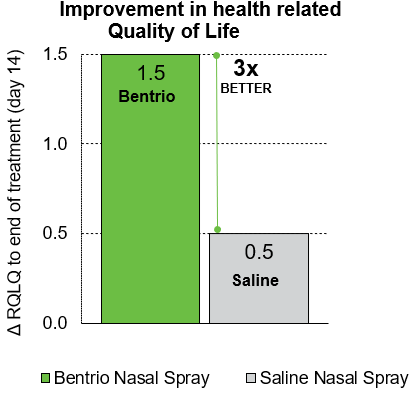
The Bentrio group in the NASAR clinical trial saw significant improvements in health-related quality of life, as measured by the Rhinoconjunctivitis Quality of Life Questionnaire (RQLQ). From the start to the end of the two-week trial, their RQLQ score improved by 1.0 points more than in the saline nasal spray group, a statistically significant and clinically relevant change. Bentrio showed significantly better outcomes across all domains of quality of life.
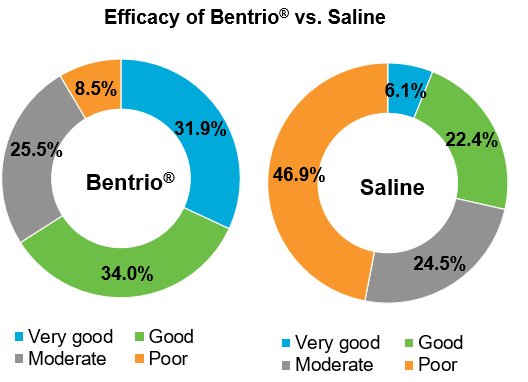
Bentrio® Nasal Spray significantly outperformed the current standard for non-medicated Allergic Rhinitis Treatment: Saline Nasal Sprays.
65.9% of Bentrio-treated study participants rated treatment efficacy as either good or very good vs. 28.5% of saline-treated participants.
Bentrio® demonstrating efficacy in a clinical trial with controlled house dust mite exposure
Significant Reduction of Increase in Total Nasal Symptom Score Due to House Dust Mite Exposure Compared to No Treatment
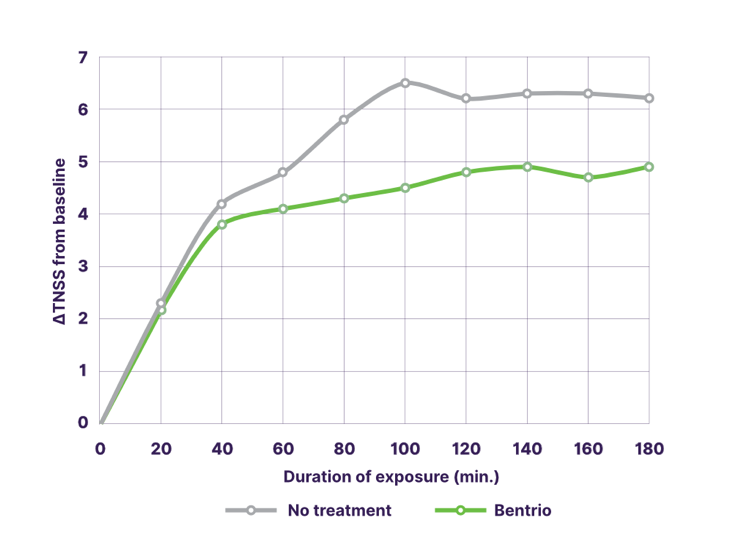
In a triple crossover trial in Canada with 37 patients suffering from perennial allergic rhinitis caused by house dust mites (HDM), Bentrio® application provided effective and statistically significant protection while exposed to HDM for 3 hours in a challenge chamber. Bentrio® treatment reduced the increase in mean TNSS by 1.1 points compared to no treatment (p<0.01; 95% confidence interval -1.87 to -0.28). Under Bentrio treatment (green line in the graph), the mean TNSS was 4.1 points vs. 5.2 points under no treatment (grey line).
Bentrio® demonstrating efficacy in a clinical trial with controlled grass pollen exposure
Significant Reduction of Increase in Total Nasal Symptom Score Due to Grass Pollen Exposure Compared to No Treatment
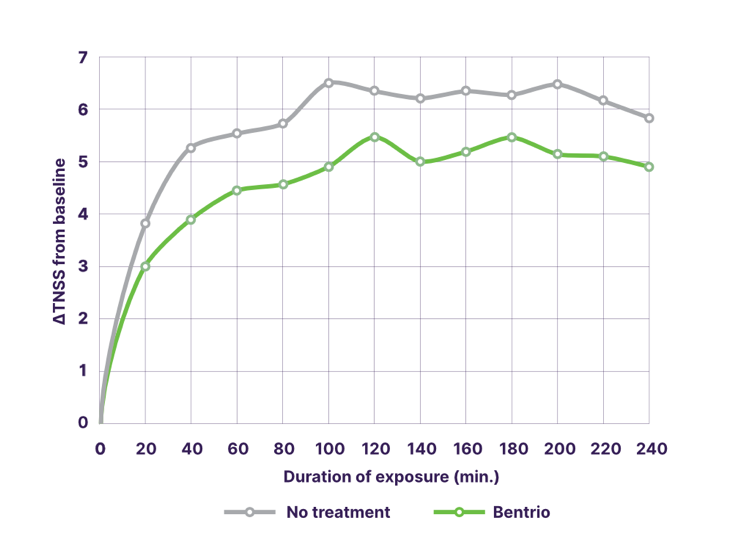
In a controlled crossover trial in Germany with 36 patients suffering from seasonal allergic rhinitis caused by grass pollen, Bentrio® application provided effective and statistically significant protection while exposed to grass pollen for 4 hours in a challenge chamber. Bentrio® treatment reduced the increase in mean TNSS by 1.09 points compared to no treatment (p<0.01; 95% confidence interval -1.61 to -0.54). Under Bentrio treatment (green line in graph), the mean TNSS was 4.82 points vs. 5.91 points under no treatment (grey line).
Bentrio® protective film covering nasal mucosa for up to 4 hours
Presence of Fluorescent Bentrio® and Saline Nasal Spray in Inferior Turbinate
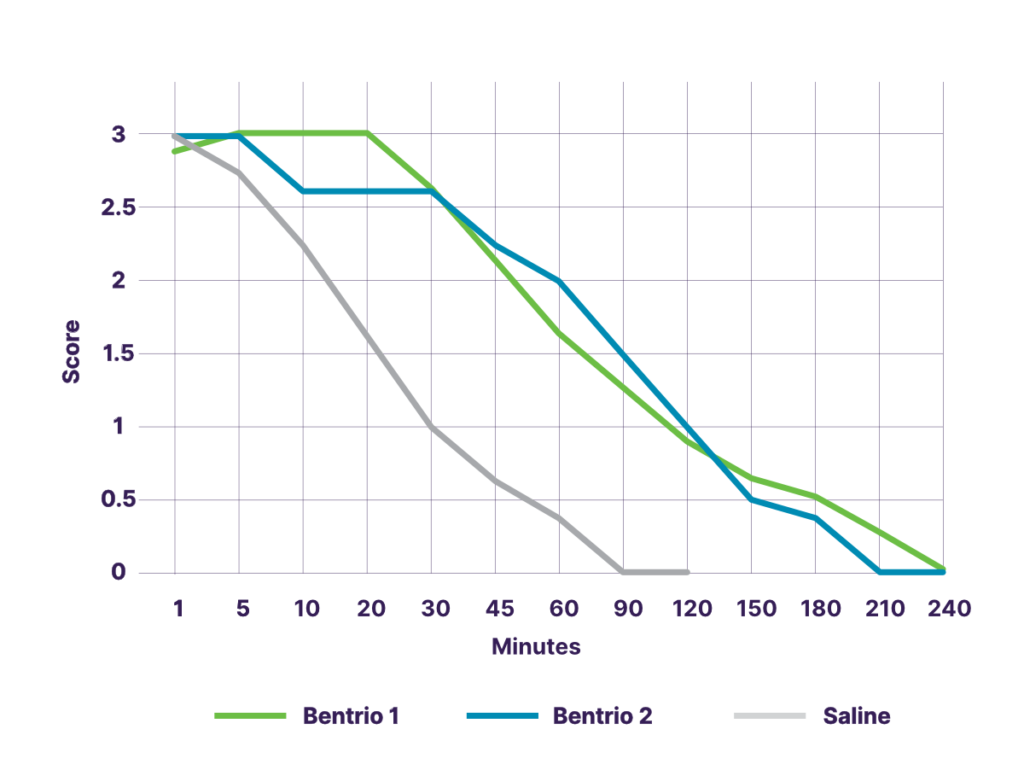
In a study with 8 human volunteers in Germany on the distribution and residence time in the nasal cavity, fluorescent Bentrio® was observed for 3.5 to 4 hours in the relevant inferior turbinate of the nose. The application of a second puff at a different angle did not show any meaningful difference, confirming that a single puff is sufficient. In contrast, fluorescent saline nasal spray was observed only for 60 minutes. The long nasal residence time of Bentrio® shown in this study corresponds well with the long duration of protection observed in the aforementioned challenge chamber studies with controlled house dust mite or grass pollen exposure.





